
Insights & Analysis
There’s one constant in healthcare: change. Count on us to break down the trends so you can stay up to date. Follow our take on each piece of this deep, intertwined, and often perplexing industry to find opportunities and practical approaches to move healthcare forward.

FDA Programs Offer Advantages for Manufacturers in Rare Disease Space
Manufacturers can accelerate innovation and improve rare and ultra-rare disease therapy access by leveraging new and established regulatory pathways.

White Paper: FQHC 340B Contract Pharmacies and Medicare HIV Drug Access
Avalere Health found that contract pharmacy relationships do not increase utilization of HIV drugs for Medicare beneficiaries with HIV receiving care at FQHCs.

Coding, Reimbursement, and Clinical Expertise to Support Market Access
Translate clinical innovation into commercial success by tapping into our market access team’s expertise in coding systems and reimbursement policies.

Over 170 Drugs May Qualify for Inclusion in the GUARD Model
Avalere Health’s analysis estimates that more than 170 drugs will be eligible for inclusion in the GUARD Model, with most concentrated in five drug categories.

Gene Therapy in Rare Disease: From Breakthrough to Stewardship
As gene therapies for rare diseases scale, system readiness and long-term stewardship–not science alone–will determine access, impact, and sustainability.

Medicare Drug Price Negotiation: Dynamics to Watch for IPAY 2028
In the IPAY 2028 negotiation cycle, stakeholders should watch how prior MFPs impact negotiations and how Part B adds new complexities.

European-level Changes for Market Access in 2026 and Beyond
Avalere Health identifies the key themes emerging from recent EU regulation that directly impacting market access.

Wider Change in the European Environment: Implications for Market Access
Avalere Health identifies EU-driven changes in 2025 that are influencing the market access environment.

USP DC 2026 Updates and Strategic USP Engagement Opportunities
The updated USP Drug Classification was recently released with several revisions to categories, classes, PGs, and products included. Beyond historical uses, USP’s classifications will be used to inform product eligibility for Medicare MFN demonstrations.

White Paper: How are Payers Thinking about Accumulators, Maximizers, and Alternative Funding Programs?
Findings from a payer survey examine trends in commercial payer use of cost avoidance strategies and how these programs could affect medication access.

CMS Announces the Third Round of Medicare Drug Price Negotiation
CMS announced the 15 drugs in Medicare Part B and D selected for negotiation for Initial Price Applicability Year 2028.

Driving Access to Rare Disease Care via Multi-stakeholder Collaboration: Perspectives from APAC Payers and HTA Experts
This third installment of our APAC rare disease series explores key enablers of achieving higher levels of multi-stakeholder collaboration in funding decisions for rare disease treatments.

Impact of US Drug Price Policy on Global Market Access
Given the uncertain challenge of future international price referencing by the US, Avalere Health considers implications for global market access.

How MFN Pricing in Part B May Affect Beneficiary OOP Costs
New analysis suggests the GLOBE Model would have limited impact on OOP costs, with over 99% of sampled Medicare Part B FFS beneficiaries seeing no reduction.
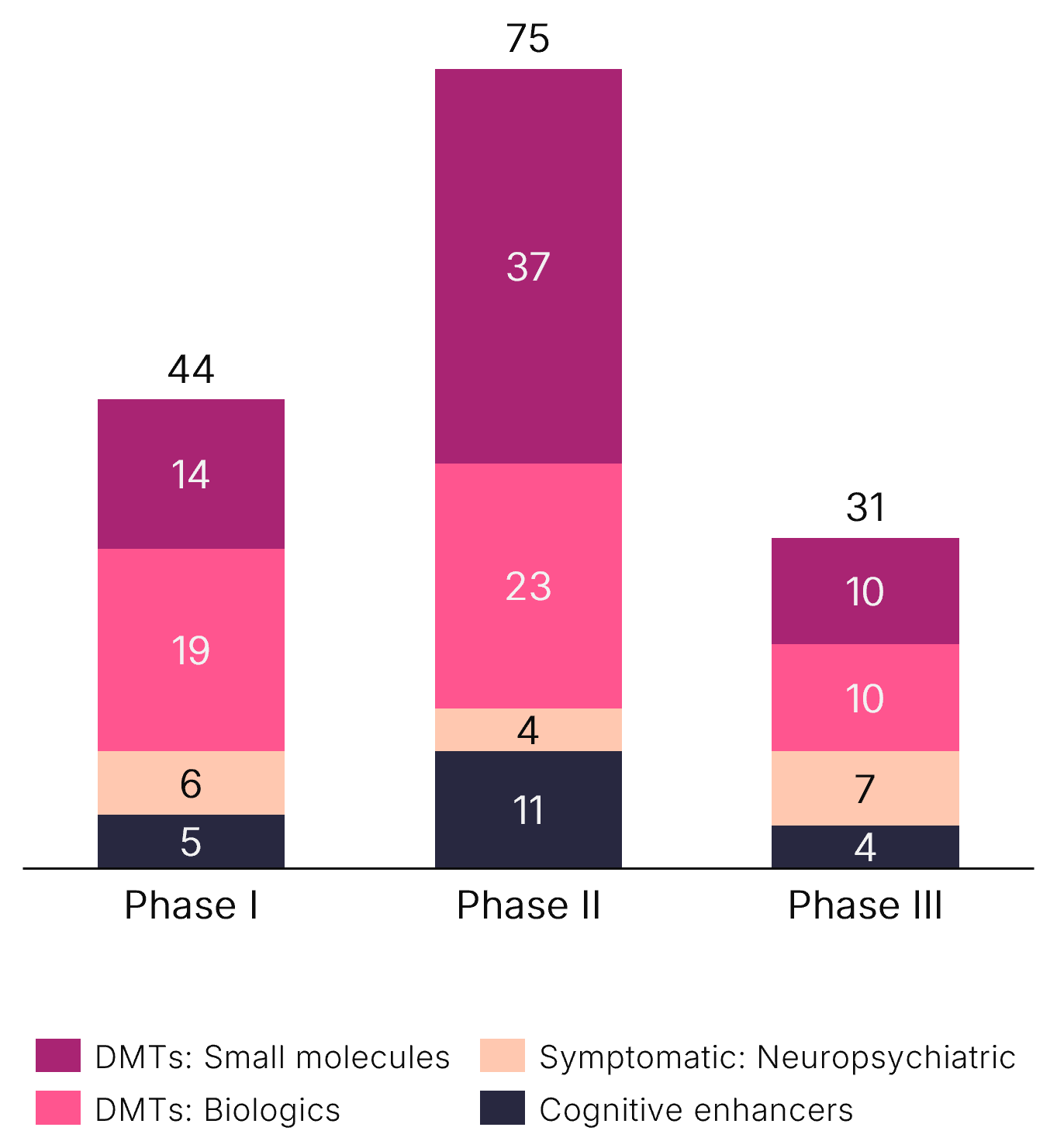
Alzheimer’s Fast-Changing Treatment Landscape and Pipeline: Need-to-Know Insights
A robust, diversified clinical development pipeline in AD requires market entrants to establish early differentiation during commercialization planning.

Now Available: 2026-2027 Coverage and Coding Regulatory Calendar
Avalere Health’s new CY 2026–2027 Coding and Regulatory Calendar supports life sciences firms’ strategic planning for critical coding deadlines, timelines, and regulatory updates.

Patients are Ready for Radioligand Therapy, But is Our Healthcare System?
Radioligand therapies show benefit across cancers, but US adoption is limited by infrastructure, reimbursement, imaging access, and care coordination gaps
Amplifying Rare Disease Patient Groups in APAC
Rare disease patient groups in APAC can amplify their impact through multi-stakeholder collaboration to achieve equitable access for people living with rare diseases.

December 2025 ACIP Insights and 2026 Emerging Priorities
The ACIP’s December meeting resulted in a key change to the pediatric immunization schedule and signaled several potential changes to US vaccine coverage and access in 2026.

340B Purchase Data Highlights Continued Program Growth
HRSA 2024 340B purchase data demonstrates continued program growth amid shifting market dynamics


