
Coverage and Payment
Coverage and reimbursement shapes patient access and affordability. Life sciences companies rely on favorable coverage decisions to ensure market access and adoption of their products, while health plans need effective reimbursement strategies to manage costs and provide comprehensive benefits to their members.


Bridging the Gap: The Future of GLP-1 Coverage in Part D
The recent delay of the BALANCE Model and the GLP-1 Bridge extension highlight the challenges of expanding GLP-1 access in Part D.

ICD-10 C&M Committee 2026 Spring Meeting: Proposed Code Revisions
On March 17-18, the ICD-10 C&M Committee discussed proposals for 35+ new codes designed to improve accuracy, address emerging health issues, and strengthen disease reporting.

Integrated Clinical and Coding Strategies Drive Market Success
Early, integrated clinical, coding, and reimbursement strategies transform market access challenges into successful launches with clear coverage and payment pathways.

How are States Responding to Medicaid Budget Pressures?
States are taking near-term Medicaid actions in response to funding pressure including changes to benefits, rates, and procurement timelines.

White Paper: Coverage and Utilization Management Trends in the Commercial Market, 2016-2025
For single-source brand drugs across eight TAs, commercial market formulary coverage, in the aggregate, declined by 20 percentage points from 2016–2025

White Paper: FQHC 340B Contract Pharmacies and Medicare HIV Drug Access
Avalere Health found that contract pharmacy relationships do not increase utilization of HIV drugs for Medicare beneficiaries with HIV receiving care at FQHCs.

Coding, Reimbursement, and Clinical Expertise to Support Market Access
Translate clinical innovation into commercial success by tapping into our market access team’s expertise in coding systems and reimbursement policies.

USP DC 2026 Updates and Strategic USP Engagement Opportunities
The updated USP Drug Classification was recently released with several revisions to categories, classes, PGs, and products included. Beyond historical uses, USP’s classifications will be used to inform product eligibility for Medicare MFN demonstrations.

Driving Access to Rare Disease Care via Multi-stakeholder Collaboration: Perspectives from APAC Payers and HTA Experts
This third installment of our APAC rare disease series explores key enablers of achieving higher levels of multi-stakeholder collaboration in funding decisions for rare disease treatments.
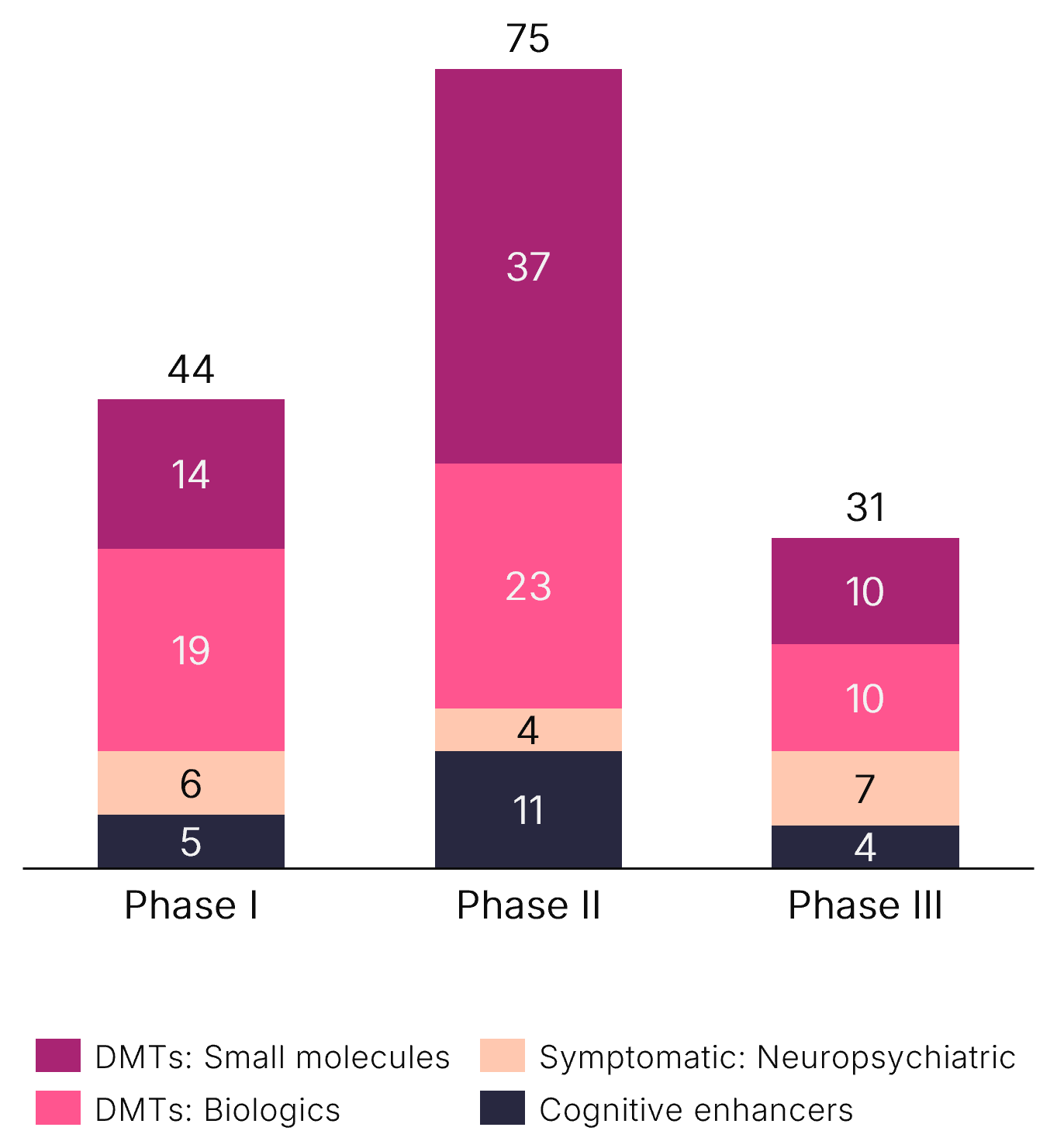
Alzheimer’s Fast-Changing Treatment Landscape and Pipeline: Need-to-Know Insights
A robust, diversified clinical development pipeline in AD requires market entrants to establish early differentiation during commercialization planning.

Now Available: 2026-2027 Coverage and Coding Regulatory Calendar
Avalere Health’s new CY 2026–2027 Coding and Regulatory Calendar supports life sciences firms’ strategic planning for critical coding deadlines, timelines, and regulatory updates.

Patients are Ready for Radioligand Therapy, But is Our Healthcare System?
Radioligand therapies show benefit across cancers, but US adoption is limited by infrastructure, reimbursement, imaging access, and care coordination gaps

State Copay Accumulator Bans Now Affect At Least 17% of Commercial Lives
State legislators are attempting to reduce patient costs and increase access through policies that block copay accumulators and maximizers.

AI Across Payers, Providers, and PBMs: Turning Pilots into Progress
Healthcare stakeholders are increasingly using AI in front- and back-office applications, but few tools have been able to achieve expected results once organizations deploy them at-scale.

Part D Formulary Management Tightens in 2026
Coverage for top brand drugs is tightening slightly in 2026, along with increased utilization management and continued shifts from copays to coinsurance.

Updated Resource: Commercial Payers’ Use of Oncology Compendia
Our updated dashboard provides insights into trends in commercial payers’ use of clinical drug compendia for off-label oncology coverage decisions.

How Can Stakeholders Engage with USPSTF Recommendation Development
The USPSTF develops independent, evidence-based preventive recommendations through an evidence-based review process. Avalere Health helps organizations successfully navigate, engage, and influence these processes.

Part D Policy Change Lowers Max OOP Costs for Many Enrollees
Cost-sharing reductions from enhanced plans and EGWPs allow beneficiaries to reach the catastrophic phase with lower OOP spending than those in basic plans.

Part D Choices Continue to Shrink with Fewer PDPs in 2026
The Part D market will contract even further in 2026, with a 22% decrease in standalone PDP options and a 9% decrease in non-SNP MA-PDs.

ICD-10 C&M Committee 2025 Fall Meeting: Proposed ICD-10-CM Code Revisions
On September 9-10, NCHS discussed proposals for 300+ new ICD-10-CM codes designed to improve accuracy, address emerging health issues, and strengthen disease reporting.


