How Can Stakeholders Engage with USPSTF Recommendation Development
Summary
The USPSTF develops independent, evidence-based preventive recommendations through an evidence-based review process. Avalere Health helps organizations successfully navigate, engage, and influence these processes.Overview
The US Preventive Services Task Force (USPSTF) is an independent panel of 16 clinical and methodological experts that issues evidence-based recommendations on clinical primary and secondary preventive services—delivered or referred from primary care settings. Services within its purview include screenings, behavioral counseling, and preventive medications.
USPSTF’s recommendations inform clinical practice, coverage determinations, and public health priorities nationwide. Under the Affordable Care Act, commercial and employer health plans must cover preventive services receiving an A or B grade from the USPSTF without cost sharing. These recommendations also inform primary care provider care management priorities.
Recent FY26 budget planning discussions within the Department of Health and Human Services (HHS) regarding potential structural or administrative changes to the USPSTF and its alignment with the Agency for Healthcare Research and Quality (AHRQ) could affect how the Task Force operates and maintains its independence. Ongoing monitoring of these developments will be essential to understanding their potential impact on evidence review processes and preventive service coverage.
As healthcare stakeholders increasingly need to engage in evidence-generation and policy shaping, understanding the USPSTF’s methods, decision making process, and opportunities for public input is essential. This Insight summarizes the USPSTF’s current approach and outlines how organizations can participate in and influence the process.
How the USPSTF Develops Recommendations
The USPSTF follows a structured, evidence-based, and iterative process to develop recommendations that balance the benefits and harms of preventive services.
1. Topic Nomination and Prioritization
Private individuals, professional societies, advocacy groups, and industry stakeholders may nominate new topics for recommendations or propose updated recommendations through the USPSTF website. Nominations are reviewed by the Task Force’s Topic Prioritization Workgroup, which assesses public health importance, relevance to asymptomatic primary care populations and settings, and availability of new evidence. Prioritized topics enter the USPSTF’s review queue yearly and undergo evidence review every three to five years.
2. Research Planning and Analytic Framework
Once a topic is prioritized, the USPSTF develops a draft research plan in partnership with AHRQ (which convenes the Task Force) and an Evidence-Based Practice Center (EPC). EPCs are overseen by AHRQ through its Effective Healthcare Program, and both operate within the U.S. Department of Health and Human Services (HHS). This plan includes an analytic framework linking preventive interventions to health outcomes, key questions (KQs) that define the chain of evidence, and contextual questions addressing implementation and public health importance. The draft research plan is posted on the USPSTF’s website for four weeks of public comment before finalization. The Trump administration’s ongoing discussions regarding the structure and funding of AHRQ underscores the importance of maintaining the USPSTF’s independence, methodological rigor, and transparency in conducting evidence reviews.
3. Systematic Evidence Review
The EPC conducts a systematic review to guide the review process, following the analytic framework and KQs developed in the research plan. This review synthesizes data on benefits, harms, and applicability to primary care populations. When direct evidence is limited, the USPSTF may use modeling studies to evaluate scenarios such as screening intervals or target populations.
4. Recommendation Development
Following review, the USPSTF evaluates adequacy and quality of evidence, magnitude of benefits and harms, and certainty of net benefit. Each recommendation receives a letter grade (A, B, C, D, or I) that reflects both the strength and certainty of evidence, from highest level of net benefit (A) to inconclusive and requiring further evidence (I).
5. Public and Expert Review
Before finalization, both the draft evidence review and draft recommendation statement are released for public comment for four weeks. Simultaneously, the USPSTF solicits review from content experts and partner organizations, including federal agencies, clinical societies, and patient groups. Comments are reviewed and considered before publication of the final recommendation statement and accompanying evidence summary.
6. Task Force Meetings
The USPSTF typically meets three times per year (March, July, and November) to review draft evidence reports, discuss methodological updates, and vote on draft and final recommendations. These meetings are closed to the public and attended by Task Force members, AHRQ staff, and invited representatives from partner federal agencies and organizations.
The July 2025 USPSTF meeting was canceled, and the November 2025 meeting was cancelled (at time of publication) due to the government shutdown. Such disruptions highlight the importance of maintaining ongoing and systematic opportunities for stakeholder engagement and transparency in the Task Force’s ongoing work. When regular meetings are delayed or cancelled, the ability to review evidence, update recommendations, and incorporate public and expert input is hindered.
Ongoing Engagement Opportunities
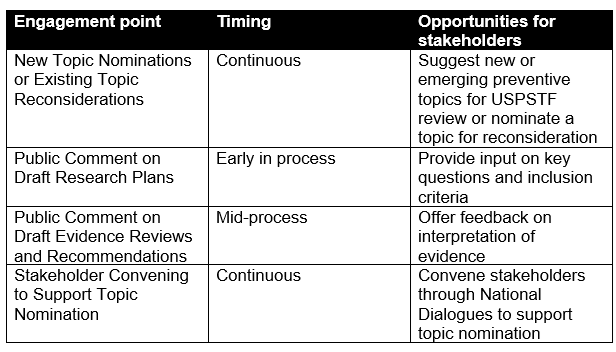
The USPSTF process offers multiple avenues for stakeholder involvement throughout the recommendation lifecycle as shown in Table 1.
Table 1. Stakeholder Engagement Opportunities with the USPSTF Process
How Avalere Health Helps Clients Like You
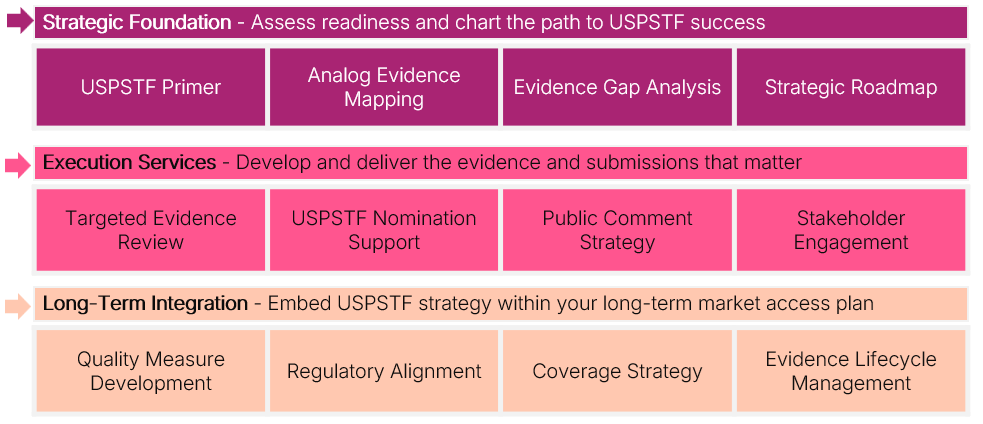
Avalere Health offers a comprehensive suite of services to guide clients through the USPSTF process, from early strategy to post-submission integration as shown in Figure 1. We begin by laying the strategic foundation: assessing USPSTF readiness through a detailed primer, benchmarking product attributes against Task Force criteria, identifying evidence gaps, and designing a tailored roadmap to align submission goals with broader access objectives.
Figure 1. Services to Guide Stakeholders from Strategy to Integration
Next Steps
Avalere Health is uniquely positioned to support innovators seeking to influence USPSTF recommendations. With deep expertise across evidence generation, regulatory strategy, public policy, and quality frameworks, we offer a cross-functional approach tailored to the nuances of preventive care. Our team has a proven track record supporting diagnostic, biopharmaceutical, and digital health companies seeking USPSTF engagement to unlock access and drive adoption. To learn more about how Avalere Health can assist you connect with us.




