Strategic Entry Points for Shaping USPSTF Recommendations
Summary
Avalere Health helps innovators shape USPSTF recommendations, driving preventive care access, payer coverage, and clinical adoption.USPSTF’s Central Role in Preventive Care Coverage
For diagnostics developers, biopharmaceutical companies, professional societies, and nonprofit organizations, engaging with the U.S. Preventive Services Task Force (USPSTF) is a critical, yet often overlooked, strategy for unlocking broad access to their preventive services. USPSTF recommendations historically and continue to play a key role in shaping coverage decisions, clinical behavior, and population health standards.
USPSTF evaluates specific preventive care interventions and issues a recommendation with a letter grade (A, B, C, D, or Insufficient [I] statement) based on the strength of existing evidence and the net benefits of a preventive service. Services that receive an “A” or “B” grade from the USPSTF must be covered by commercial and public insurers without cost sharing, per the Affordable Care Act. These grades also drive clinical decision support systems, inform population health quality measures, and influence payer coverage and reimbursement policies. Because many primary care providers closely follow USPSTF recommendations, alignment with these guidelines is often essential for optimal uptake in the clinical setting.
Despite the significance of USPSTF grades, many evidence-based clinical offerings—particularly diagnostics and digital technologies—struggle to achieve meaningful adoption over the product lifecycle without a deliberate USPSTF engagement strategy.
Two Strategic Entry Points: New Topic Nomination and Existing Topic Reconsideration
Two primary entry points for engaging with USPSTF are new topic nomination and existing topic reconsideration. Stakeholders with robust clinical or real-world evidence, particularly those addressing under-recognized populations or conditions, have a unique opportunity to shape national preventive policy and drive long-term adoption.
A new topic nomination is a formal proposal for the USPSTF to evaluate a new primary or secondary preventive service. Any individual or organization (for example manufacturers, clinicians, professional societies, etc.) can nominate a service for USPSTF consideration provided it includes an asymptomatic population, is provided in or referable from the primary care setting, has a strong data-backed rationale for public health importance, and has the potential to meaningfully affect clinical practice. Submission of a new topic to USPSTF is particularly valuable for organizations introducing innovations that address gaps in early detection or prevention. The Task Force website states that “the USPSTF generally has selected one or two new topics a year for which to evaluate the evidence and develop a recommendation.” Working with a partner with both deep expertise in developing a compelling submission and navigating consideration by the Task Force is key.
Reconsideration of an existing topic is a process by which a stakeholder requests an update to an existing USPSTF recommendation based on new evidence. This route is ideal for technologies or services that previously received an “I” (insufficient evidence) or “D” (not recommended) grade, and for which significant new data may change the net benefit assessment. Reconsideration requests require a focused and evidence-driven strategy, demonstrating that updated research findings could alter the Task Force’s grade or clinical guidance. Once again, the selection of an expert and seasoned partner provides greater opportunities for successful reconsideration.
End-to-End Support for USPSTF Engagement
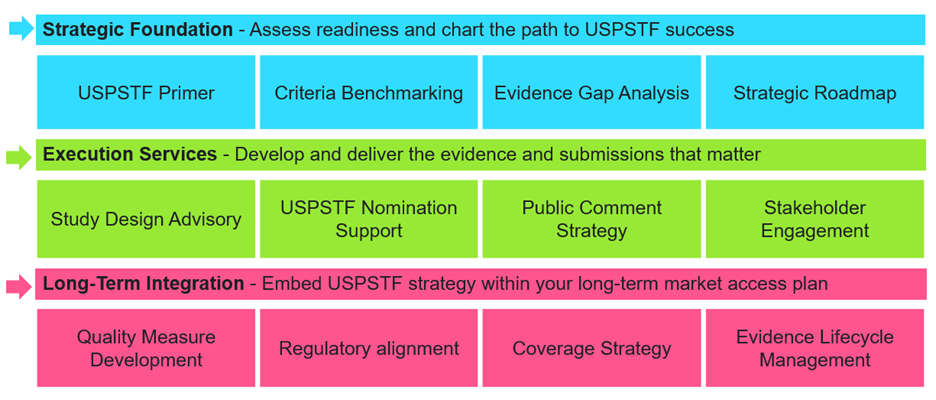
Avalere Health offers a comprehensive suite of services to guide clients through the USPSTF process, from early strategy to post-submission integration as shown in Figure 1. We begin by laying the strategic foundation: assessing USPSTF readiness through a detailed primer, benchmarking product attributes against Task Force criteria, identifying evidence gaps, and designing a tailored roadmap to align submission goals with broader access objectives.
From concept to decision, we manage every stage of the nomination or reconsideration pathway, delivering study-design guidance, hands-on USPSTF submission support, and strategic advice on framing evidence for maximum public health impact. Our team provides study design advisory, USPSTF nomination submission support, and expert guidance on positioning evidence for public health impact. We also lead stakeholder engagement, public comment strategies, and alignment with key partners to maximize visibility and support.
Once a submission is underway or accepted, we help clients sustain momentum by embedding USPSTF strategy into long-term market access and policy planning. This includes contributions to quality measure development, navigating regulatory and payer pathways, and managing the lifecycle of supporting evidence. By linking USPSTF strategy with broader access and uptake initiatives, we ensure that preventive innovations not only gain adoption, but also achieve preventive health impact.
Figure 1. Services to Guide Stakeholders from Strategy to Integration
Next Steps
Avalere Health is uniquely positioned to support innovators seeking to influence USPSTF recommendations. With deep expertise across evidence generation, regulatory strategy, public policy, and quality frameworks, we offer a cross-functional approach tailored to the nuances of preventive care. Our team has a proven track record supporting diagnostic, biopharmaceutical, and digital health companies seeking USPSTF engagement to unlock access and drive adoption. To learn more about how Avalere Health can assist you connect with us.




