Gene Therapy in Rare Disease: From Breakthrough to Stewardship
Summary
As gene therapies for rare diseases scale, system readiness and long-term stewardship–not science alone–will determine access, impact, and sustainability.The Gene Therapy Landscape in Rare Disease is Maturing
Gene therapies for rare diseases are moving from isolated, one-off interventions to an established treatment modality with significant implications for healthcare delivery and system design.
By the end of 2025, the US Food and Drug Administration (FDA) had approved 26 gene therapies across in vivo, ex vivo, and cell-based platforms. After early, sporadic introductions, the period from 2022 to 2023 marked a clear inflection point, with multiple modalities simultaneously entering clinical practice. While activity appeared to slow in 2024, this reflects regulatory timing rather than a contraction in development momentum; 2025 signaled renewed activity across platforms and indications.
The majority of these gene therapies address rare or ultra-rare conditions, with each modality bringing distinct requirements for manufacturing, delivery, monitoring, and follow-up, thus increasing operational complexity as portfolios expand. This raises a core question for the rare disease sector: Can infrastructures built for scarcity sustain delivery, evidence generation, and oversight at scale?
Figure 1. FDA-approved Gene Therapies by Modality and Year (2015–2025)
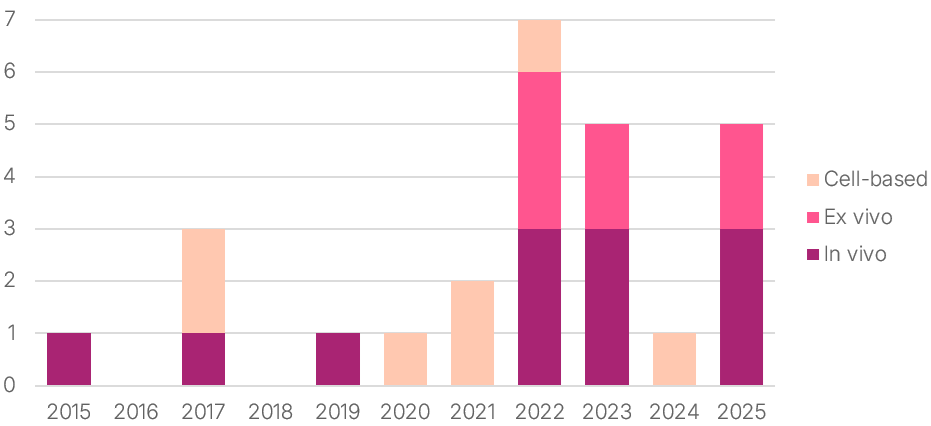
Note: Excludes non–gene-modified cell therapies.
Importantly, these therapies do not enter a neutral landscape. Rare diseases already carry substantial societal and economic burden. In the United States alone, the aggregate economic burden of rare diseases has been estimated at nearly $1 trillion annually when accounting for direct medical costs and indirect productivity losses across just 379 analysed diseases. Despite recent advances, fewer than 5% of rare diseases have an approved therapy, leaving the vast majority of conditions without effective treatment options.
Temporal Mismatch: Annual Systems, Multi-decade Obligations
Gene therapies are typically administered once, yet their benefits, risks, and evidentiary obligations unfold over many years. By contrast, healthcare systems operate on annual budgets, short coverage horizons, and near-term evidence expectations.
In the rare disease space, this misalignment is particularly acute. Prolonged diagnostic odyssey, narrow referral pathways, and concentrated specialist expertise already strain delivery. One-time interventions therefore collide with systems built for chronic management, creating pressure for redesigned care pathways, sustained monitoring infrastructure, and continuity of evidence over time.
This persistence is not discretionary. For many gene therapies, regulators require structured, long-term follow-up extending up to 15 years to monitor delayed safety signals and durability of effect. Delivery thus initiates an extended regulatory and operational commitment that sits uneasily alongside short-term funding cycles and fragmented accountability. The result is not simply a pricing challenge, but a structural tension between how gene therapies function and how systems are financed and governed.
Ownership Mismatch: Fragmented Accountability over Time
Responsibility for long-term outcomes is fragmented. Manufacturers may scale back involvement once post-approval commitments are met. Payers change as patients move across plans or geographies. Providers deliver a one-time intervention but are expected to monitor outcomes for years. Patients themselves bear potentially irreversible biological and financial risk. No single stakeholder experiences the full spectrum of benefits, costs, and accountability.
Rare disease ecosystems further complicate this picture by relying on non-commercial actors. Patient organizations, advocacy groups, and philanthropic entities have long been the driving force behind rare disease research prioritization, trial recruitment, and data generation. As gene therapies scale, these actors are increasingly drawn into post-market evidence generation and access discussions, often without clear governance structures, defined ownership, or sustained resourcing. Patients, caregivers, and patient advocacy groups should be involved in clinical development programs, regulatory conversations, commercialization phases, and post-marketing approval. Early and often engagement of these stakeholders at each phase of the drug development program increases chances of success.
Portfolio Effect: When Exceptions Accumulate
Health systems can accommodate isolated exceptional therapies, but struggle when exceptions accumulate.
As developers build gene therapy portfolios, system impact becomes cumulative rather than product-specific. Infrastructure designed for scarcity, such as specialist expertise concentrated in centers of excellence, must now support multiple therapies in parallel. Requirements for delivery, monitoring, and long-term follow-up therefore stack across assets, rather than resetting after each launch.
Rare diseases have long functioned as proving grounds for innovation, driving advances in platform technologies, regulatory flexibility, and patient engagement models that later diffuse into broader medicine. As portfolios expand, however, gene therapy developers’ role increasingly shifts from scientific experimentation to operational orchestration: coordinating shared infrastructure, aligning follow-up expectations, and sequencing launches to avoid system overload.
Without deliberate, portfolio-level planning, these cumulative demands risk constraining uptake, slowing access, and ultimately limiting the scalability of gene therapy innovation in the rare disease ecosystem.
Strategic Implications for Gene Therapy Developers in Rare Diseases
The shift from breakthrough interventions to enduring commitments carries clear implications for developers operating in the rare disease space:
- Stewardship must be designed, not assumed: Long-term evidence generation, follow-up models, and data governance increasingly shape access and system confidence and must be considered and embedded early.
- Portfolio strategy is now a system strategy: As post-treatment delivery obligations accumulate across assets, launch sequencing, shared infrastructure, and capacity planning become central to sustainable growth.
- Access strategy must account for ill-defined ownership: Payer churn and diffuse accountability weaken incentives for improved access to these treatments unless addressed through aligned evidence and strong partnership models.
- Trust is becoming a differentiator: In the rare disease sector, where communities are tight-knit, and histories of unmet needs are long, sustained credibility with patients, regulators, and providers increasingly depends on developers’ ability to plan for and support ongoing system responsibility.
Avalere Health supports a spectrum of stakeholders across the highly complex rare disease ecosystem in assessing system in assessing system readiness, quantifying long-term stewardship obligations, and designing access and evidence strategies aligned with the evolving realities of gene therapy portfolios.
To learn more, connect with us.

