Over 170 Drugs May Qualify for Inclusion in the GUARD Model
Summary
Avalere Health’s analysis estimates that more than 170 drugs will be eligible for inclusion in the GUARD Model, with most concentrated in five drug categories.Background
In December 2025, the CMS Innovation Center (CMMI) introduced the proposed Guarding U.S. Medicare Against Rising Drug Costs (GUARD) Model as part of ongoing efforts to advance Most-Favored Nation (MFN) pricing in the US. MFN pricing aims to align US drug prices with those paid in economically comparable countries.
As proposed, the model is mandatory for manufacturers with eligible drugs and its performance period is January 1, 2027 through December 31, 2031. The model would apply to a randomly selected 25% sample of geographic areas to represent 25% of beneficiaries in standalone Prescription Drug Plans and Medicare Advantage Prescription Drug plans, excluding beneficiaries in Employer Group Waiver Plans.
For each eligible GUARD Model drug, CMS will calculate a model-specific rebate, which will be equal to the difference between a Part D drug’s Medicare net price and an international benchmark amount. The GUARD rebate will also factor in any inflation rebates owed by the manufacturer.
Eligibility Criteria for GUARD Model Drugs
Drugs eligible for inclusion in the GUARD Model must:
- Be single-source Part D drugs eligible for inflation rebate reporting
- Have at least $69 million in gross Part D drug spending over a 12-month period
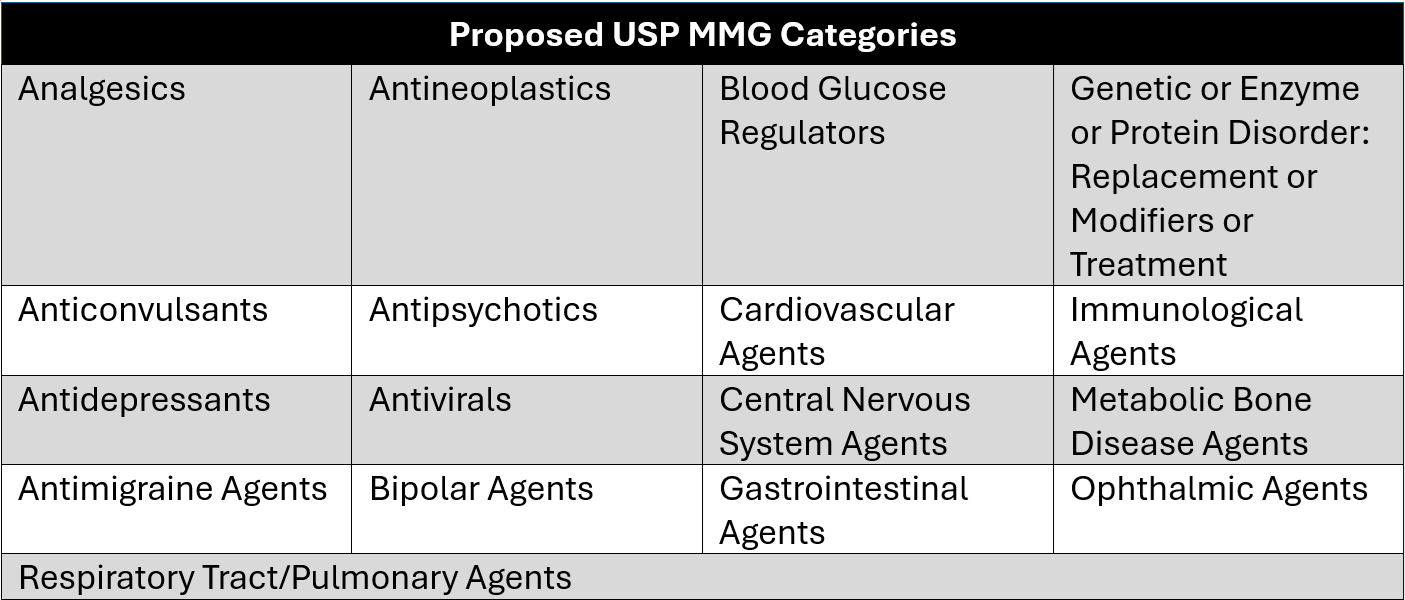
- Fall into one of 17 select United States Pharmacopeia (USP) Medicare Model Guidelines (MMG) categories (Table 1).
Table 1: Proposed USP MMG Categories for GUARD Model Eligibility
Drugs with an effective maximum fair price (MFP) in effect under the Medicare Drug Price Negotiation Program will be excluded from GUARD.
To evaluate the impact of the GUARD Model, Avalere Health estimated both the number of Part D drugs that could be eligible for inclusion and the proportion of total Part D spending attributable to these drugs.
Findings
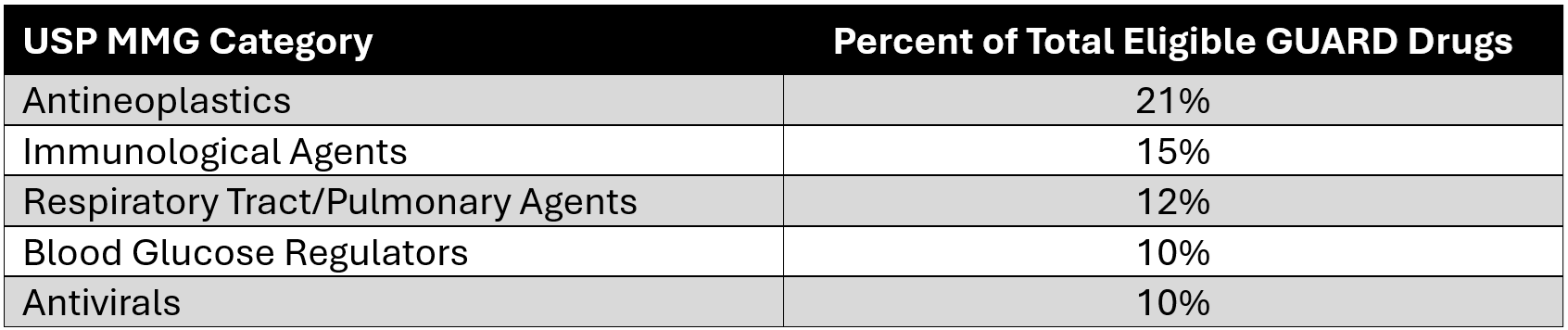
Based on the proposed GUARD Model drug eligibility criteria and 2024 gross Part D drug spending, Avalere Health estimates that over 170 drugs would qualify for inclusion in GUARD at the start of the model. These drugs account for almost $93 billion in total gross Part D spending, representing approximately 32% of all Part D drug spending in 2024. The majority of drugs that would qualify for inclusion fall under one of five USP MMG categories: Antineoplastics, Blood Glucose Regulators, Immunological Agents, Respiratory Tract/Pulmonary Agents, and Antivirals (Table 2).
Table 2: Percentage of GUARD Model-Eligible Drugs in Top 5 USP MMG Categories
While the model is designed to include approximately 25% of Part D beneficiaries through random sampling of geographic areas—which CMS notes will account for about 25% of Part D spending—the impact may differ by drug. For some products, the share of volume captured by the model may be higher or lower than 25% of the beneficiary population taking the drug. This deviation is more likely for products that treat smaller patient populations that may be unevenly distributed across regions.
Eligibility for the GUARD Model is based on several factors; however, final eligibility will depend in part on whether a drug falls into one of the 17 USP MMG categories from the most recent USP MMG available. USP is an independent organization that creates classification systems that health plans can leverage to create and maintain formularies for drugs. Historically, every three years, USP updates its Medicare Model Guidelines, which help determine how drugs are grouped and classified under Medicare. The draft of the next version of these guidelines is expected to be released in June 2026 for a month-long comment and consultation period and finalized in September, ahead of the proposed January 2027 model start date. If finalized this year, updates to classifications of products in the USP MMG could impact which drugs are eligible for inclusion in the GUARD Model.
Collaborate with Avalere Health’s Experts for Strategic USP and MFN Pricing Model Support
As new payment and international reference pricing models take shape, USP categorization can directly influence drug eligibility for the GUARD and GLOBE Models. Avalere Health’s USP and drug pricing experts partner with clients to navigate USP classification updates, identify strategic opportunities, and analyze product and portfolio implications of these and other drug pricing policy changes on volume, pricing, competitive positioning, and patient access dynamics.
To learn more about how Avalere Health can support your strategic planning around MFN drug pricing policies, connect with us.
Methodology
Avalere Health used 2024 Part D claims data released by CMS and applied GUARD Model drug eligibility parameters. However, GUARD model eligibility will ultimately be determined based on gross drug spend from the corresponding performance year (e.g., 2027 gross drug spend for performance year 2027); therefore, any changes in drug spend between 2024 and 2027 may affect final model eligibility. Additionally, Avalere Health’s estimation of GUARD model eligible drugs relies on current single-source status; if a currently single-source drug faces generic or biosimilar competition prior to the start of the model, it may be excluded from the GUARD eligible drug list. While there is uncertainty about how the GUARD Model will interact with other models, such as the GENErating cost Reductions fOR U.S. Medicaid (GENEROUS) Model and other MFN deals with the administration, this analysis assumes that all drugs that meet the GUARD Model eligibility criteria would be included in the model.




