Direct to Consumer Drug Purchasing: What Comes Next
Summary
The growth of DTC drug purchasing programs may shift the patient access and affordability landscape, with implications for manufacturers, payers, and other stakeholders.Rapid Grown of DTC Programs
Even though a small subset of companies have introduced targeted direct-to-consumer (DTC) purchasing pilots for the last couple of years, these programs have grown significantly in size and scale since the Trump administration released an Executive Order (EO) in May and sent letters on Most-Favored Nation (MFN) pricing to a select set of manufacturers in July. The EO and letters encourage the use of DTC purchasing programs as a strategy to lower drug costs and tackle what the administration sees as broader drug pricing inefficiencies within the current system.
As of October 22, 10 manufacturers have launched DTC programs for select products, though only 3 have explicitly tied their actions to the administration’s broader drug pricing efforts. In mid-October, the Trump administration announced the establishment of TrumpRx, a public website that will aggregate in-market DTC programs for participating manufacturers.
New Avenues for Patient Access, Affordability, and Data Collection
DTC programs offer patients a user-friendly interface that allows them to “shop” for affordable prices for medications. These platforms may offer telehealth services, prescription management, and fulfillment/delivery. Overall, they place more decision-making power in the hands of the patient, and have the potential to disrupt the current chain of drug supply, payment, and access. Additionally, DTC programs allow for the collection of robust patient decision-making data that may not be available via traditional channels.
Key Considerations for Manufacturers in DTC Program Design
The Trump administration’s EO and letters did not dictate DTC program structure and design, which has led to a wide range of DTC offerings and considerations, including:
- Cash-pay vs. insurance integration: DTC programs may operate inside or outside of a patient’s insurance (the latter is typically referred to as cash-pay). To date, many DTC programs have prioritized cash-pay, which focus on clear and transparent pricing, but may not be a viable option for many patients. Future expansions of insurance-integrated programs could expand affordability and enable wider patient uptake. Integration with insurance programs will also allow for tracking of primary and secondary adherence, Prescription Drug Event claims capture, ensure reduction in patient gap in care and interact with Stars another measures of quality.
- Product selection: Certain products may be better suited for DTC sales, particularly for cash-pay programs. In selecting products to make available on these platforms, manufacturers should consider the coverage landscape, patient demand, and product pricing and price elasticity vs. inelasticity.
- Program design and operationalization: DTC programs often leverage third-party vendors to design front- and back-end program interfaces, disease education, telehealth, product distribution, and pharmacy fulfillment. By investing in DTC platforms, manufacturers can glean critical insights into patient behavior and adherence, which may otherwise be unavailable in a fragmented supply chain. Assessing DTC vendor capabilities across these functions will be critical to balance sustainability with agility to meet evolving needs of patients, providers and payers.
Next Steps for Stakeholders Amid DTC Program Growth
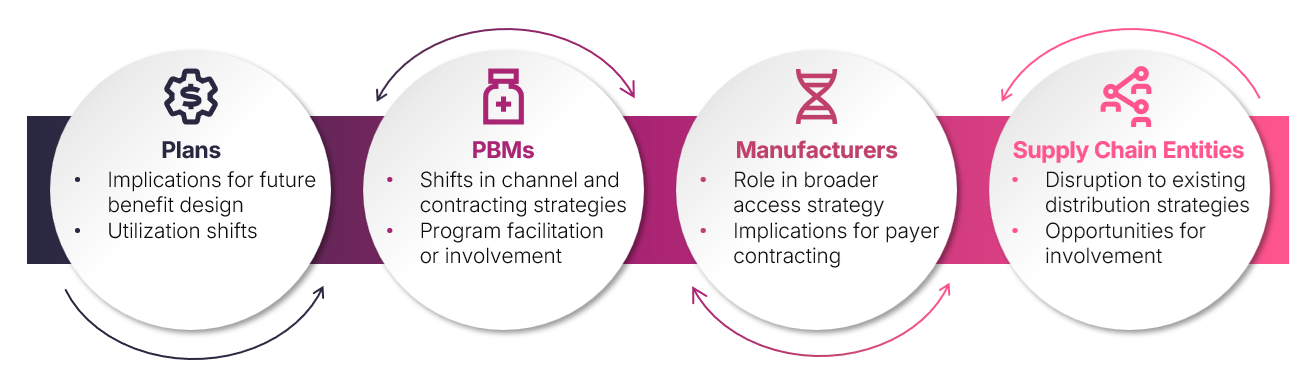
DTC programs present an alternative pathway to access and fulfillment, creating potential disruption and opportunity for manufacturers, payers, and other supply chain stakeholders. These trends are also surfacing questions around the complex interplay of issues like the role of prescribers, patient monitoring, consumer demand, anti-kickback considerations, existing patient support services and a range of potential spillover effects. As these programs grow, stakeholders can consider opportunities for involvement and intersections with broader business:
Avalere Health Can Support Stakeholders Navigating Uncertainty
The drug pricing and access landscape continues to evolve as policy changes such as MFN, the Inflation Reduction Act, and the One Big Beautiful Bill Act create ripple effects.
Avalere Health’s seasoned experts in market access, US and global pricing strategy, and federal and state policy support organizations with a range of tailored activities, including:
- Manufacturer portfolio and pipeline assessment
- Landscape of DTC vendor and manufacturer offerings
- Assessment of DTC approaches and scenario planning
- Projected shifts to pharmaceutical supply chain and measurement of impact to abandonment, adherence and persistence rates
- Support for federal policy design and engagement
To learn how Avalere Health can support, connect with us.




