Alzheimer’s Treatment Has Changed. Has Value?
Summary
Emerging evidence in Alzheimer's disease highlights a gap between measured effects and lived experience, shaping how value is defined, evidenced, and assessed.A New Treatment Era and a New Set of Questions
Treatment for Alzheimer’s disease (AD) is entering a new phase. After decades of stalled progress, two new disease-modifying therapies have emerged. Anti-amyloid monoclonal antibodies (mAbs), lecanemab and donanemab, have received regulatory approvals in the US and several jurisdictions in Europe, the Middle East, Africa, and the Asia-Pacific region, with additional reviews ongoing globally. For the first time, therapies that target the underlying biology of the disease rather than managing symptoms alone are now available.
Yet regulatory clearance has not translated into uniform patient access, hindering the ability of these newly approved therapies from reaching every patient possible. Around the world, payers, health technology assessment (HTA) bodies, clinicians, patient groups, and the research community remain divided on a deceptively simple question: What does value look like in Alzheimer’s disease?
The stakes go well beyond these two products. With more than 100 pipeline agents in clinical development, spanning anti-amyloid, anti-tau, neuroinflammation, and combination approaches, the frameworks used to define, substantiate, and assess value in AD today will shape the terms of access for the next generation of therapies.
The Gap Between Effect and Experience
Clinical trials for lecanemab and donanemab demonstrate statistically significant slowing of cognitive and functional decline versus placebo, and both therapies effectively clear amyloid from the brain. However, the magnitude of benefit observed on standard assessment scales has become a point of contention. On the Alzheimer’s Disease Assessment Scale–Cognitive Subscale (ADAS-Cog) cognitive subscale, treatment differences at 18 months from baseline have been characterized by some as modest, raising the question of whether patients and families would notice them in everyday life.
This tension is underscored by a recent meta-analysis of 17 randomized controlled trials involving more than 20,000 participants. The review concluded that the effects of anti-amyloid mAbs on cognition and daily functioning were “trivial” to “small at best,” and that successful amyloid clearance did not appear to produce clinically meaningful improvements. The review also highlighted increased risks of amyloid-related imaging abnormalities (ARIA), including brain swelling and microhaemorrhage.
The response from the clinical community and industry was swift and polarised. Critics of the review argued that it did not sufficiently distinguish between earlier unsuccessful anti-amyloid agents and newer therapies with demonstrated efficacy, potentially understating recent progress. Others interpreted the findings as further evidence that, despite decades of investment, the amyloid hypothesis has yet to deliver meaningful impact on patients’ lived experience.
Overall, cross-trial comparisons are inherently limited by differences in design, populations, and endpoints. However, to date no individual trial has shown the incremental benefit on the Clinical Dementia Rating–Sum of Boxes (CDR-SB) exceeding the 1.0-point MCID threshold (Figure 1).
Figure 1: Treatment Effect (CDR-SB vs. Placebo at 18 Months) Across Anti-amyloid Trials
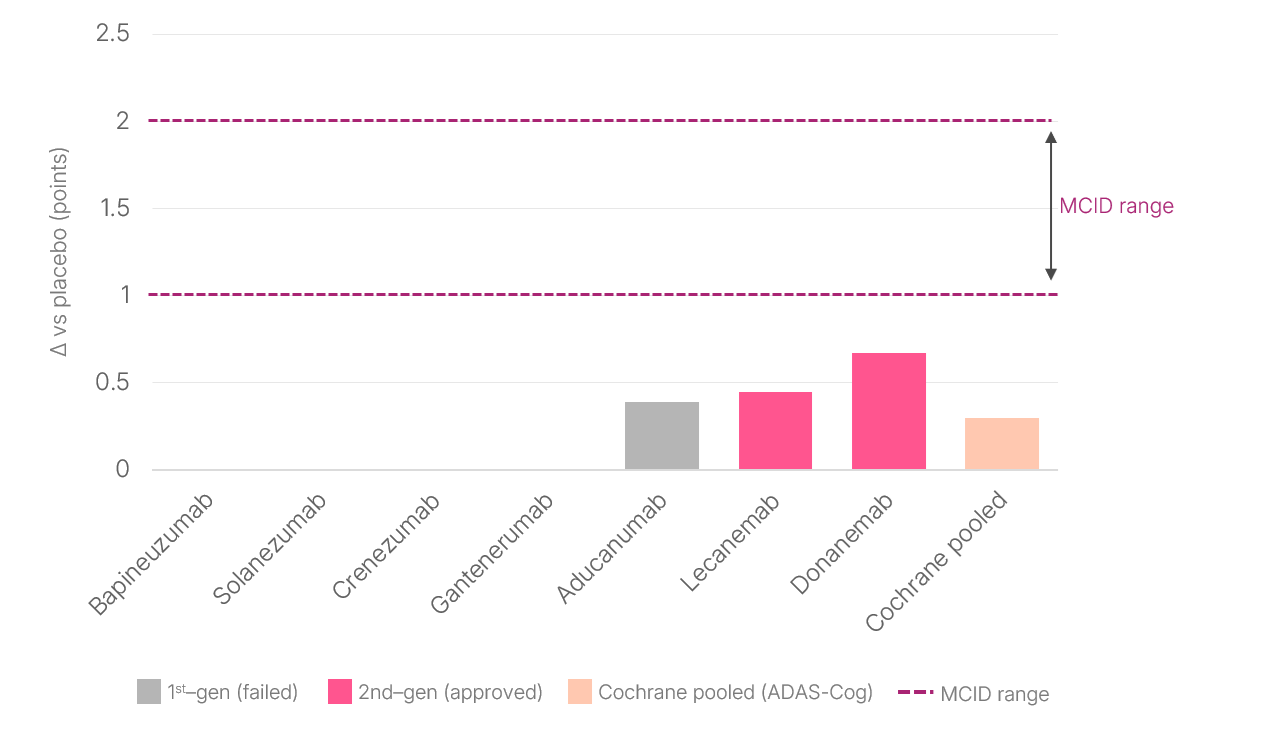
Note: Range between dotted lines marks the commonly cited Minimal Clinically Important Difference (MCID) (1.0-2.0 points)
For those developing, pricing, and seeking access to therapies for the treatment of AD, the practical implication is clear: Statistical significance on a clinical scale that falls below established meaningfulness thresholds will be challenged by HTA bodies and will not position a product well in pricing negotiations. In fact, it may result in reimbursement being denied entirely. Payers, HTA bodies, and—increasingly—regulators are demanding evidence that connects biological effects to the outcomes patients, caregivers, and health systems actually experience.
Defining Value: What Assessors Are Struggling With
The challenge of defining value in AD is playing out most visibly within the UK’s National Institute for Health and Care Excellence (NICE), which has now rejected lecanemab and donanemab on three separate occasions, citing clinical benefits too modest to justify their total cost, including infusion delivery, MRI monitoring, and management of ARIA.
Both manufacturers successfully appealed in January 2026 on two key grounds: insufficient time to respond to NHS England’s infusion cost estimates and, more importantly, concerns about the committee’s treatment of carer utility values. Evidence suggests that standard instruments such as the EQ-5D substantially underestimate the burden on unpaid caregivers. NICE has since reopened consultation, with a decision expected later in 2026.
This issue goes to the heart of how value is assessed. AD (and other forms of dementia) imposes burdens that are disproportionately borne by informal carers, partners, and family members providing sustained supervision and support, yet their experience is only partially captured in conventional quality-of-life measures. The appeal outcome signals a shift toward recognizing caregiver impact as integral, rather than secondary, to value assessments in dementia.
In the US, regulatory approvals have also been accompanied by cautious uptake. In 2022, the Centers for Medicare and Medicaid Services issued a National Coverage Determination for these therapies, but adoption has been constrained by infrastructure requirements, prior authorization, and payer-specific criteria. Parallel assessments by the Institute for Clinical and Economic Review suggested that the therapies could be cost-effective only at significantly lower price points than listed – a position that increasingly shapes commercial payer negotiations.
Across the world, the core tension is the same: Existing HTA frameworks rely on metrics that do not fully capture the societal burden of AD, including the toll on unpaid caregivers and indirect healthcare costs such as delayed institutionalization, emergency admissions, long-term care expenditure, and lost productivity. Nor are they well-suited to a disease in which therapeutic benefit may accrue over years rather than within an 18-month trial window.
Demonstrating Value: Where the Gaps Are
If defining value in AD is contested, so is demonstrating it. Several structural challenges cut across markets globally:
- Short trial windows for a long disease. Eighteen-month trial durations provide a limited view of a disease that unfolds over decades. Payers, regulators, and HTA bodies in high-income countries are all calling for long-term durability data, yet the infrastructure for sustained follow-up (registries, real-world evidence platforms, interoperable health records) remains broadly underdeveloped.
- Outcome measures designed for later-stage disease. The CDR-SB and ADAS-Cog were developed decades ago. While validated and widely used, they may compress the earliest functional losses (e.g., managing finances, navigating appointments, maintaining social connections) into aggregate scores that obscure their significance for patients at the preclinical disease stages.
- Caregiver impact is recognized but structurally underweighted. The burden on unpaid caregivers, including lost productivity, foregone career progression, and reduced societal participation, is widely acknowledged in AD research. Yet value assessments remain anchored to direct patient medical costs and clinical symptom scales rather than long-term cost offsets or indirect costs. HTA bodies have historically given limited formal weight to carer-specific evidence, thereby reducing the incentive for developers to generate it. The NICE appeal outcome may shift this dynamic: The finding that the committee’s handling of carer utility was unreasonable signals that, in the absence of carer impact data, it is now a material vulnerability in the value case, not just an evidence gap.
What This Means for Those Navigating the AD Landscape
The AD landscape is reminding the pharmaceutical industry that value is a negotiated construct shaped by evidence, appropriately selected endpoints, stakeholder perspectives, and healthcare system readiness (Figure 2).
For developers, market access teams, and evidence generation leaders operating in this space, several priorities emerge:
- Invest in broader, meaningful evidence from the outset. Clinical endpoints must be supplemented with outcomes that reflect functional impact, patient experience, and caregiver burden. Evidence-generation strategies should be stratified by stakeholder type and resonate with all stakeholders.
- Build for long-term evidence generation. Registries, real-world evidence platforms, and post-market studies that address durability, heterogeneity of response, and health-system impact over time are becoming prerequisites for sustained market access across geographic lines.
- Engage with the evolution of value assessment frameworks. HTA methodologies are evolving, albeit unevenly, through mechanisms such as severity modifiers, caregiver utility inclusion, and broader societal perspectives. There are precedents in oncology and rare diseases, where modifiers are anchored to quantitative clinical endpoints. In AD, where meaningful benefit is defined by function, independence, and caregiver burden, these dimensions will need to be more explicitly reflected within value assessment frameworks.
- Approach access as a system-level challenge. Pricing and reimbursement are shaped by a broader ecosystem of diagnostic infrastructure, workforce capacity, and pathway design, all of which fundamentally differ across markets. The access strategy must account for all these elements and be both locally tailored and globally coordinated.
- Monitor and prepare for a rapidly evolving landscape. With over 30 therapies for the treatment of AD in late-stage development, new data and regulatory decisions will accumulate quickly. Structures that enable evidence and access strategies to adapt in near real time are essential.
Figure 2: Dimensions of Value in AD
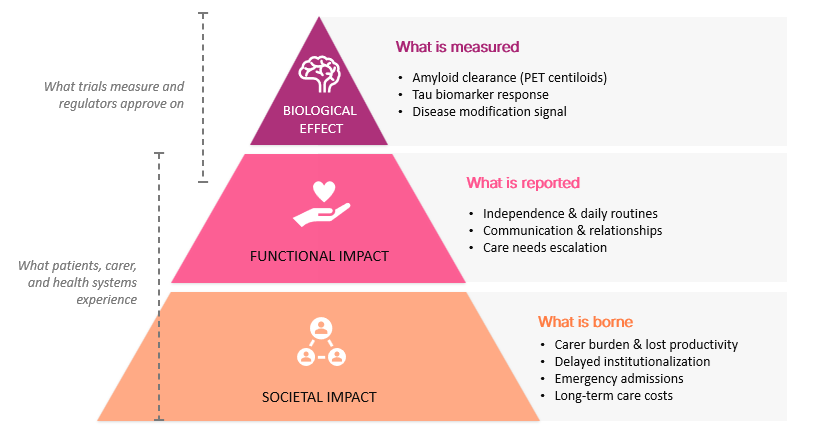
Source: Avalere Health framework; illustrative representation.
Avalere Health works with stakeholders across the AD ecosystem, from early-stage development through to post-market evidence and access, to navigate the evolving value, evidence, and assessment landscape to ensure that treatments reach EVERY PATIENT POSSIBLE.
Our teams bring deep, cross-market expertise in health economics and outcomes research, HTA engagement, patient-centered evidence generation, and market access strategy across the US and globally. To learn more about how we can support your work, connect with us.




