Inpatient Hospital Changes Proposed for Cell Therapy
Summary
CMS proposes a CAR-T payment increase in FY 2027, mediated by proposed changes to the outlier payment.Proposed Changes for CAR-T Cases
In the fiscal year (FY) 2027 Inpatient Prospective Payment System (IPPS) proposed rule, the Centers for Medicare & Medicaid Services (CMS) outlined several updates that would impact provider reimbursement for chimeric antigen receptor T-cell (CAR-T) and other cell and gene therapies (CGTs). Per the rule, CMS proposes to use its Medicare Severity Diagnosis-Related Group (MS-DRG) for CAR-T treatment stays, but with a 7% increase in the base rate.
The rule also proposes a 14% increase to reimbursement rates for cases that use CAR-T treatments as part of a clinical trial. These proposed increases would be countered by a proposed increase to the threshold for outlier payment. The financial impacts of these provisions will vary by hospitals, dependent on key geographic differences and hospital billing practices.
Background
Since the first Food & Drug Administration approval of a CAR-T product in 2017, stakeholder concerns have persisted over how the Medicare program would reimburse for these products. CAR-T therapies are commonly administered in the inpatient setting and have a significant cost for providers (e.g., average sales prices [ASPs] exceeding $450,000). Hospital inpatient reimbursement is calculated on a case-by-case basis using an MS-DRG base payment rate that is adjusted for factors such as hospital geography, diagnosis, case severity, and discharge status. Additional reimbursement can be provided through new technology add-on payments (NTAPs) and outlier payments.
For FY 2026, inpatient stays with CAR-T treatment are currently assigned to MS-DRG 018, which has a base reimbursement rate of $314,231. Outlier payments are available to hospitals to cover extremely costly cases when the costs exceed the total of the MS-DRG payment, the NTAP amount (if applicable), and the current fixed-loss threshold of $40,397. Even with these adjustments, Medicare reimbursement for CAR-T cases today sometimes fails to cover total hospital costs, which can negatively impact provider uptake and patient access.
Payment Changes for CAR-T Cases
Due to proposed increases in the base operating and capital rates for all IPPS payments and an increase in the proposed relative weight for MS-DRG 018, the proposed base payment for CAR-T cases in FY 2027 would increase by 6.9%, to $335,904.
High-Cost Outlier Payments
The proposed fixed-loss threshold for FY 2027 is $51,704, a 28% increase over the current threshold. For CAR-T cases, which are more likely than other inpatient stays to qualify for outlier payments, fluctuations in the threshold are particularly important, as they dictate the losses hospitals must incur before qualifying for an outlier payment. The combination of an increased base rate for CAR-T cases and a higher outlier threshold will counter one another and reimbursement will likely remain stable, perhaps with a small decrease for cases that rely on outlier reimbursement (see Figure 1).
Adjustment for Clinical Trial Cases
CMS reimburses CAR-T clinical trial cases, which do not incur drug costs, at a lower rate than non-clinical trial cases. Using its standard approach, CMS proposed an adjustment factor of 0.17 to the relative weight of MS-DRG 018 in FY 2027, for a base rate of $57,104. This would be a 13.6% increase over the current clinical trial payment rate (using an adjuster of 0.16). Therefore, if finalized, hospitals will receive higher payments for CAR-T cases when provided as part of a clinical trial at the beginning of the new fiscal year.
Figure 1. Illustrative Example of Hospital Reimbursement for CAR-T Cases Under IPPS in Final FY 2026 vs. Proposed FY 2027 Rule
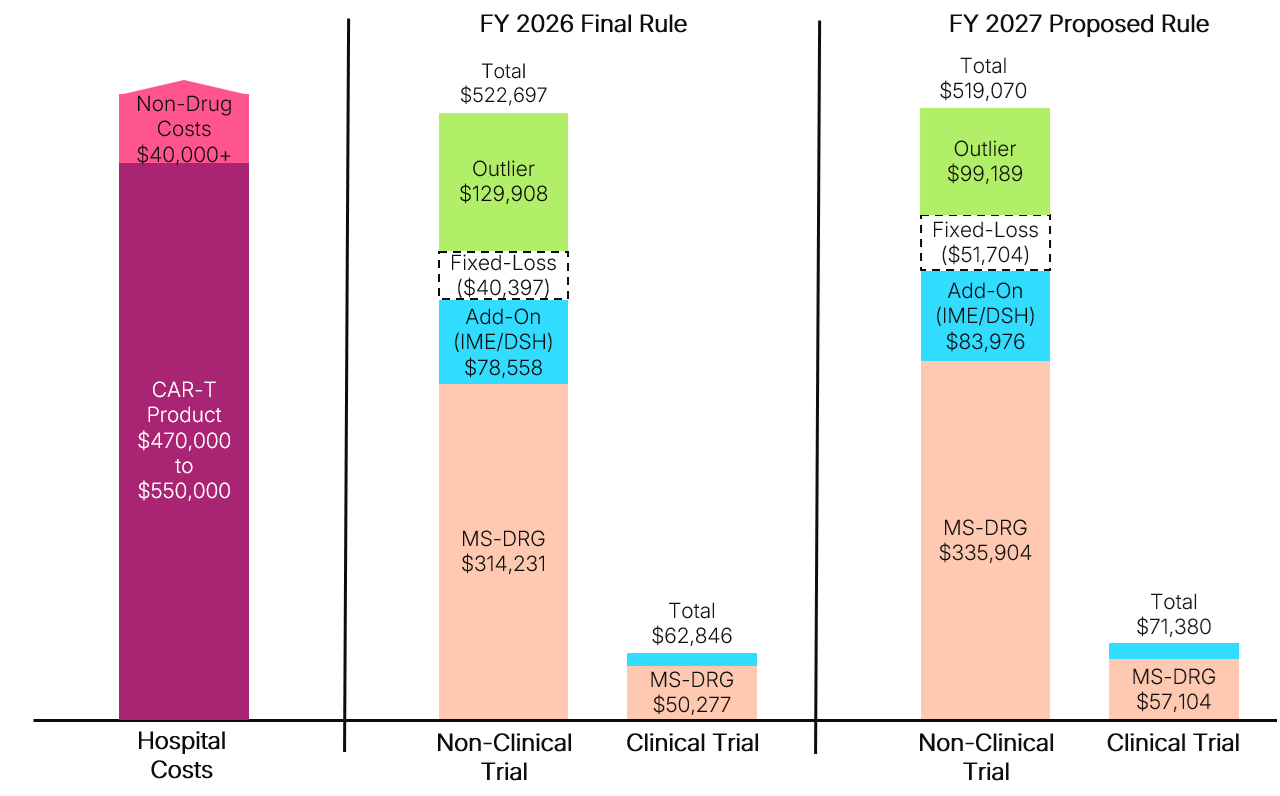 IME: Indirect Medical Education; DSH: Disproportionate Share Hospital; Note: See Appendix for assumptions made in this example.
IME: Indirect Medical Education; DSH: Disproportionate Share Hospital; Note: See Appendix for assumptions made in this example.
Product NTAP Assessments
CMS is evaluating several CGT products for NTAP in FY 2027 and requests public comments on whether each product meets newness, cost, and clinical improvement criteria required for NTAP status.
The CGT products being considered include:
- Orca-T, an allogeneic stem cell and T-cell immunotherapy for treatment of acute myeloid leukemia, acute lymphoblastic leukemia, high-risk myelodysplastic syndrome, and mixed-phenotype acute leukemia.
- Etuvetidigene autotemcel (WASKYRA™), a cell-based autologous gene therapy for the treatment of Wiskott-Aldrich Syndrome. CMS proposes not to approve NTAP.
- Prademagene zamikeracel (ZEVASKYN™), an autologous cell sheet-based gene therapy for the treatment of recessive dystrophic epidermolysis bullosa. CMS proposes not to approve NTAP.
Key Considerations
Stakeholders should consider several potential impacts that could stem from the proposed FY 2027 changes for existing assets and for future CGTs.
- Stability of MS-DRG 018: Profitability by case will become increasingly variable, given the continuing assignment of therapies to the MS-DRG. CMS noted this dynamic in the proposed rule, stating “we are in the process of carefully considering the feedback we have previously received about ways in which we can continue to appropriately reflect resource utilization associated with cell and gene therapies while maintaining clinical coherence and stability in the relative weights under the IPPS MS–DRGs.” Specifically, additional immunotherapies mapped to the MS-DRG can lead to fluctuations in the base rate and could eventually lead CMS to consider splitting the MS-DRG depending on the number of cases and differences in resource costs.
- NTAP Eligibility: As the number of CAR-T products available grows, it may be increasingly difficult for new products to clearly satisfy criteria for newness, cost, and clinical improvement relative to on-market products.
- Market-Based MS-DRG Payment Rates: The calendar year 2026 Outpatient Prospective Payment System (OPPS) rule finalized a policy use median payer-specific Medicare Advantage negotiated rates for MS-DRGs to set the relative weights for IPPS fee-for-service (FFS) cases starting in FY 2029. This data collection requirement became effective for cost reporting periods ending on or after January 1, 2026, and could be impactful for CAR-T, where negotiated rates for MS-DRG 018 may differ from the current base rates available under the FFS methodology and influence future relative weights. In the OPPS final rule, CMS also noted that there is currently no plan for a “phase-in” of the revised relative weights but that it would be considered in future rulemaking. The FY 2027 IPPS proposed rule makes no mention of the changes outlined in the CY 2026 OPPS rule.
- Site of Care Shifts: Differences in Medicare reimbursement methodology for the inpatient versus outpatient setting can often result in higher reimbursement for CAR-T in the outpatient setting, where CAR-Ts are typically separately paid at ASP+6%. While most cases are currently provided in the inpatient setting, the removal of Risk Evaluation and Mitigation Strategy requirements for CAR-T products could increase outpatient use. Additionally, new accreditation pathways for community-based physicians could allow provider office treatment when clinically supported.
Next Steps
Carefully monitoring reimbursement for these innovative products will allow CGT manufacturers, providers, and payers to engage other stakeholders based on anticipated developments. To discuss how Avalere Health can support your business on issues related to commercialization, NTAP proposal submissions, provider reimbursement, or policy developments, connect with us.
Appendix
- Hospital charges for CAR-T episode are kept constant across all examples, consistent with the geometric mean charges included in the FY 2027 Proposed Rule after outliers removed file ($1,985,237)
- Hospital has an average operating and capital cost-to-charge ratio of 0.3, which influences CMS calculations of hospital costs.
- Hospital receives add-on payments that stem from payments for IME and DSH adjustments, which are calculated as a percentage of the base MS-DRG. In this figure, there is an assumed IME factor of 0.2 and DSH adjustment of 0.05
- Hospital area wage index is 1.0, meaning no adjustment is made to the labor-related share of the standardized amount.
- Hospital received outlier payment, which is only made once hospital losses exceed the fixed-loss amount. Outlier payments provide 80% of excess costs beyond this threshold.




